Project details
Description
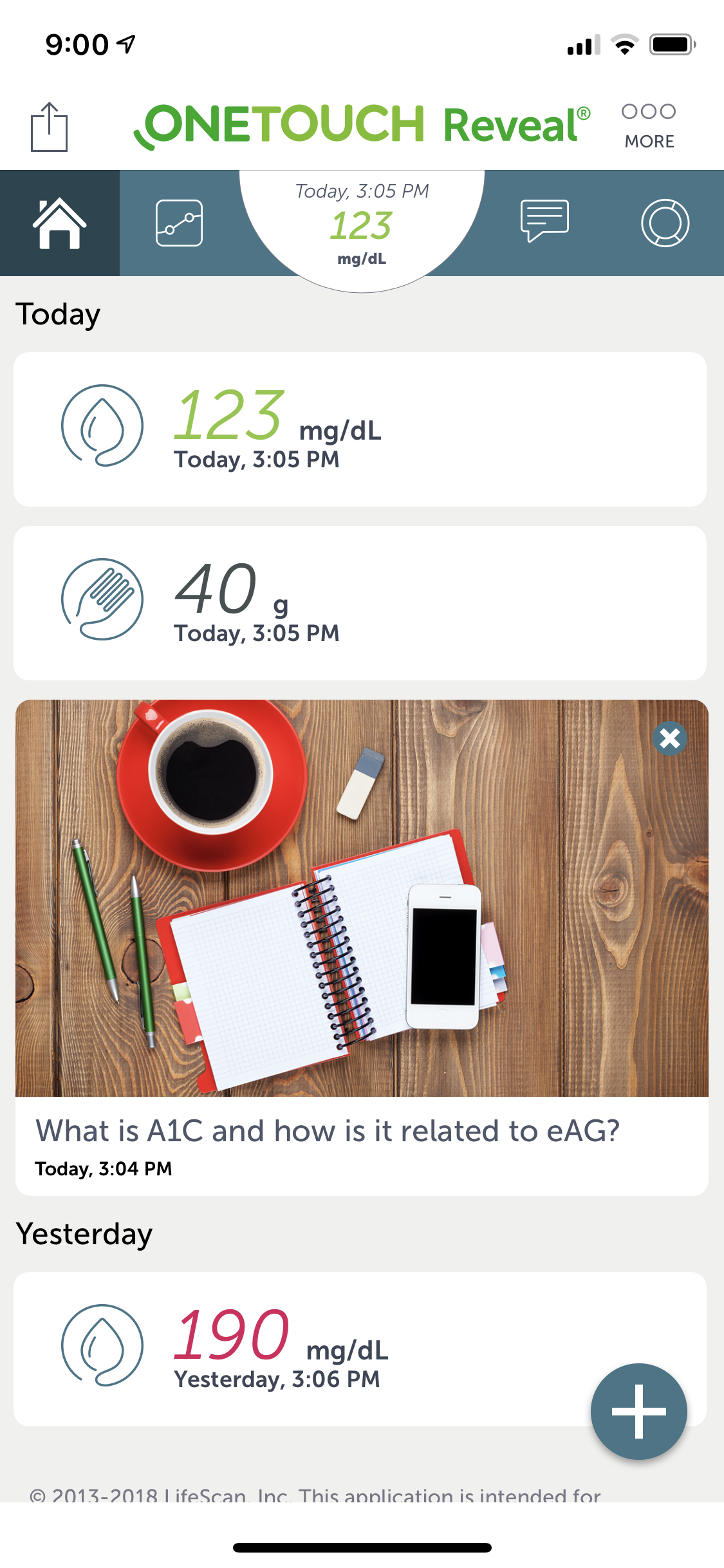
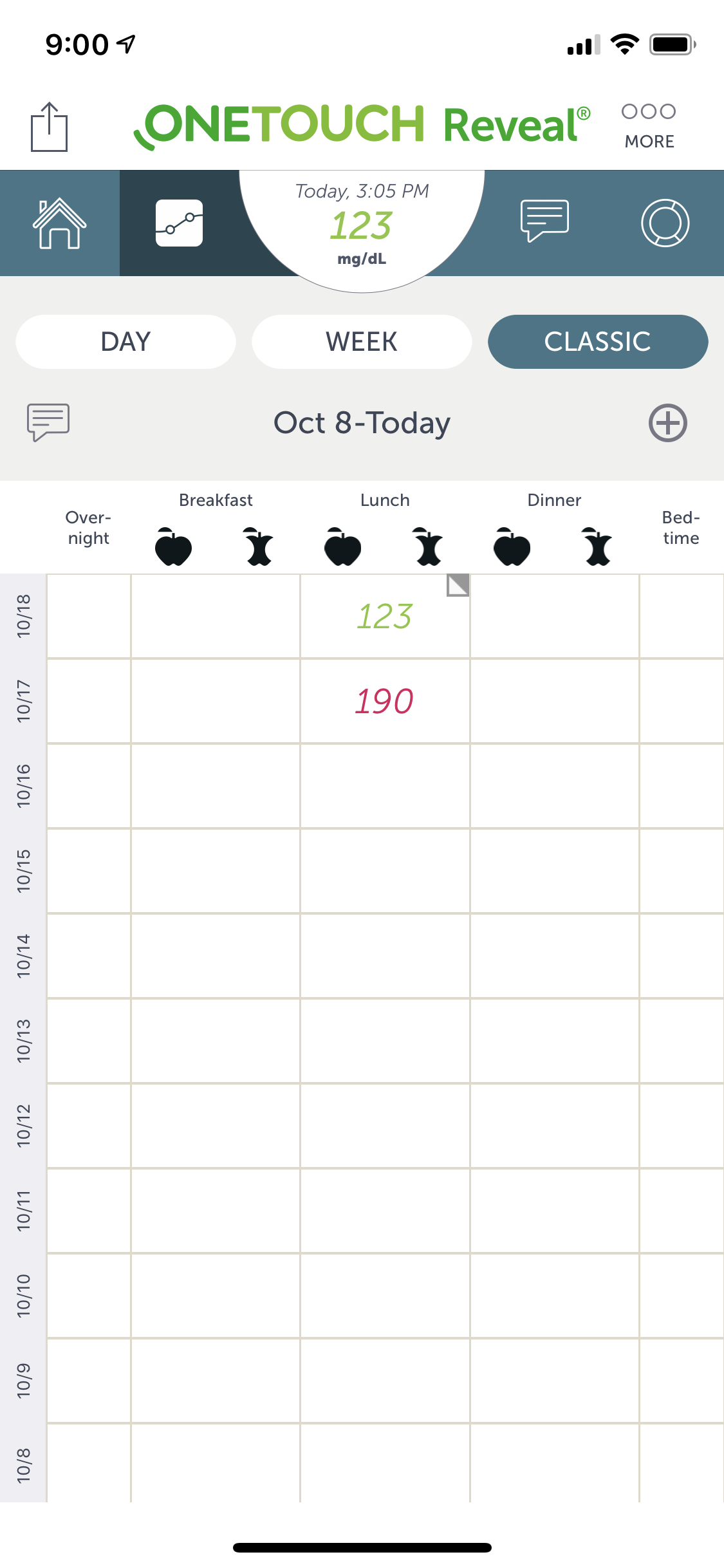
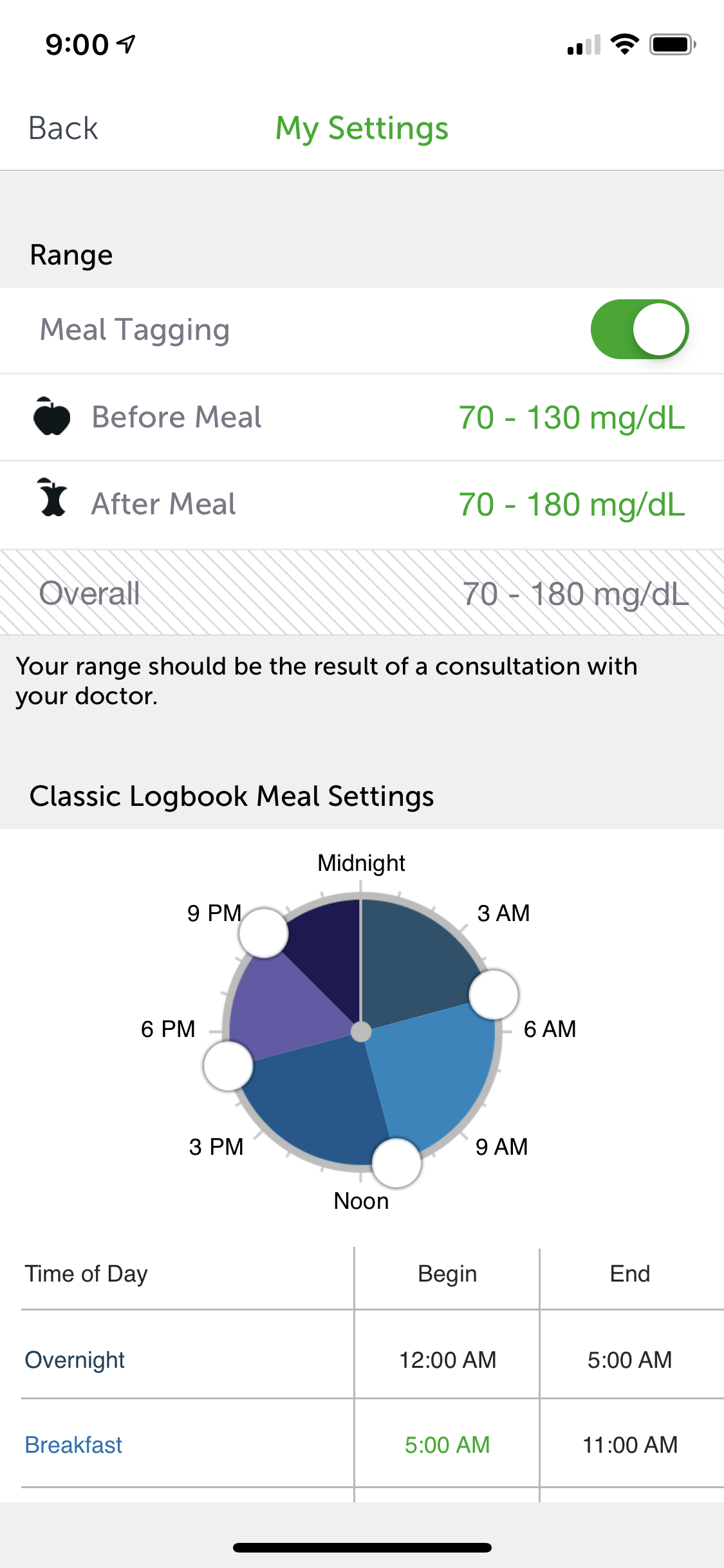
Working on J&J's LifeScan team, I was brought on to rapidly create designs and prototypes for their OneTouch Reveal mobile diabetes support application. As the project lead, I quickly acclimated and started producing. Day-to-day I worked with extremely talented team members.
-
Start Date:
April 2018 -
Final Date:
October 2018 -
Status:
Completed -
Client:
Johnson & Johnson -
Location:
King of Prussia, Pennsylvania
AT A GLANCE
Features
- Smartphone and smartwatch app availability
- Back-end HCP reporting and monitoring
- Detailed FDA 510k submission
- User tested analysis and strategy
Benefits
- Accessible and custom glucose monitoring
- Government regulated and compliant
- Professional aesthetics including white-labeling
- Integrated information aggregation and dashboard analysis
SOLUTIONS
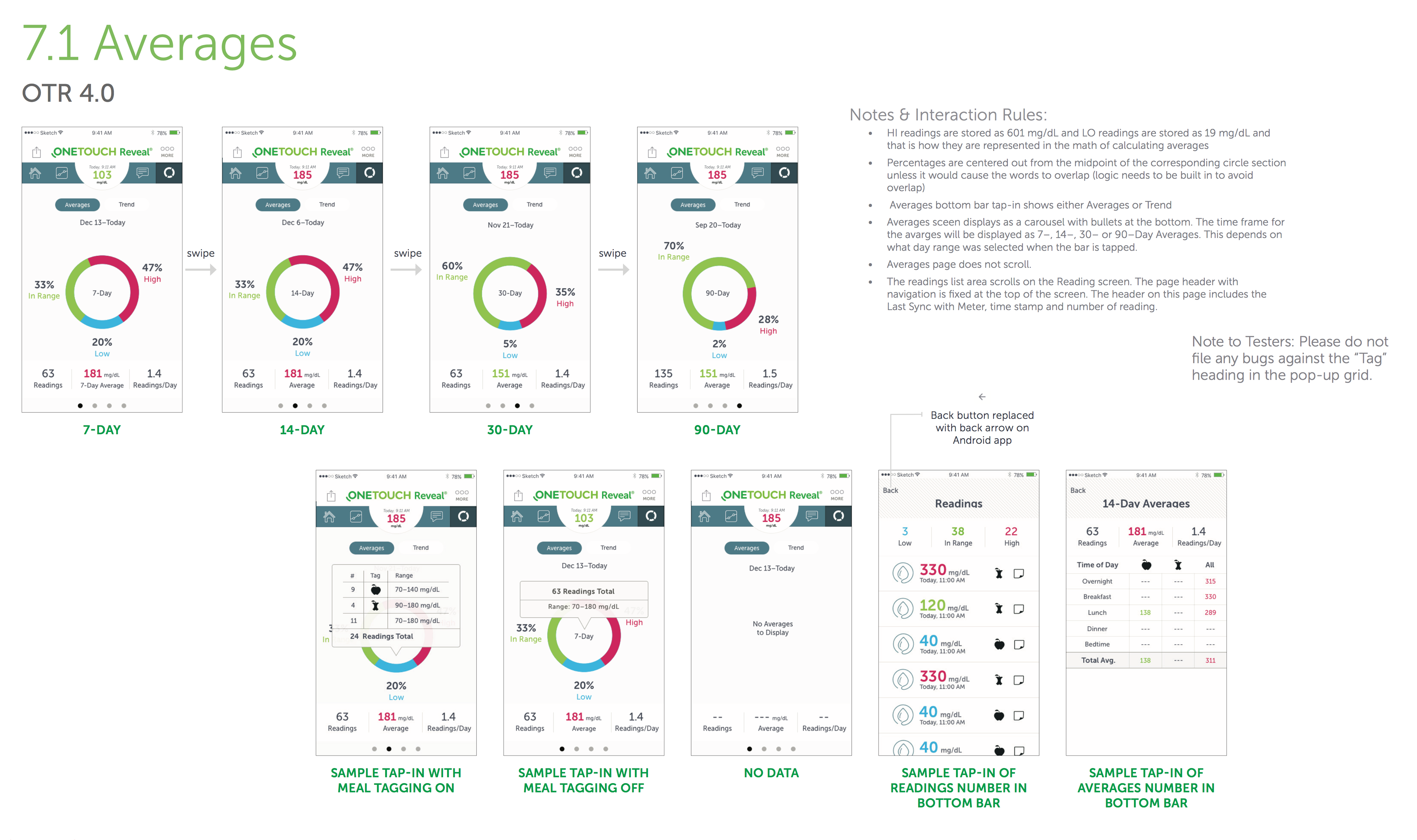
- Created mobile platform wireframes, flows, and designs in Sketch, Photoshop and Illustrator
- Consulted on design strategies with regards to FDA 510k medical device submissions and approval
- Used Axure RP 8 and Flinto to craft high-fidelity, interactive prototypes for mobile device user testing
- Consulted on areas needing WCAG accessibility attention and provided solutions for ADA compliance
OUTCOMES
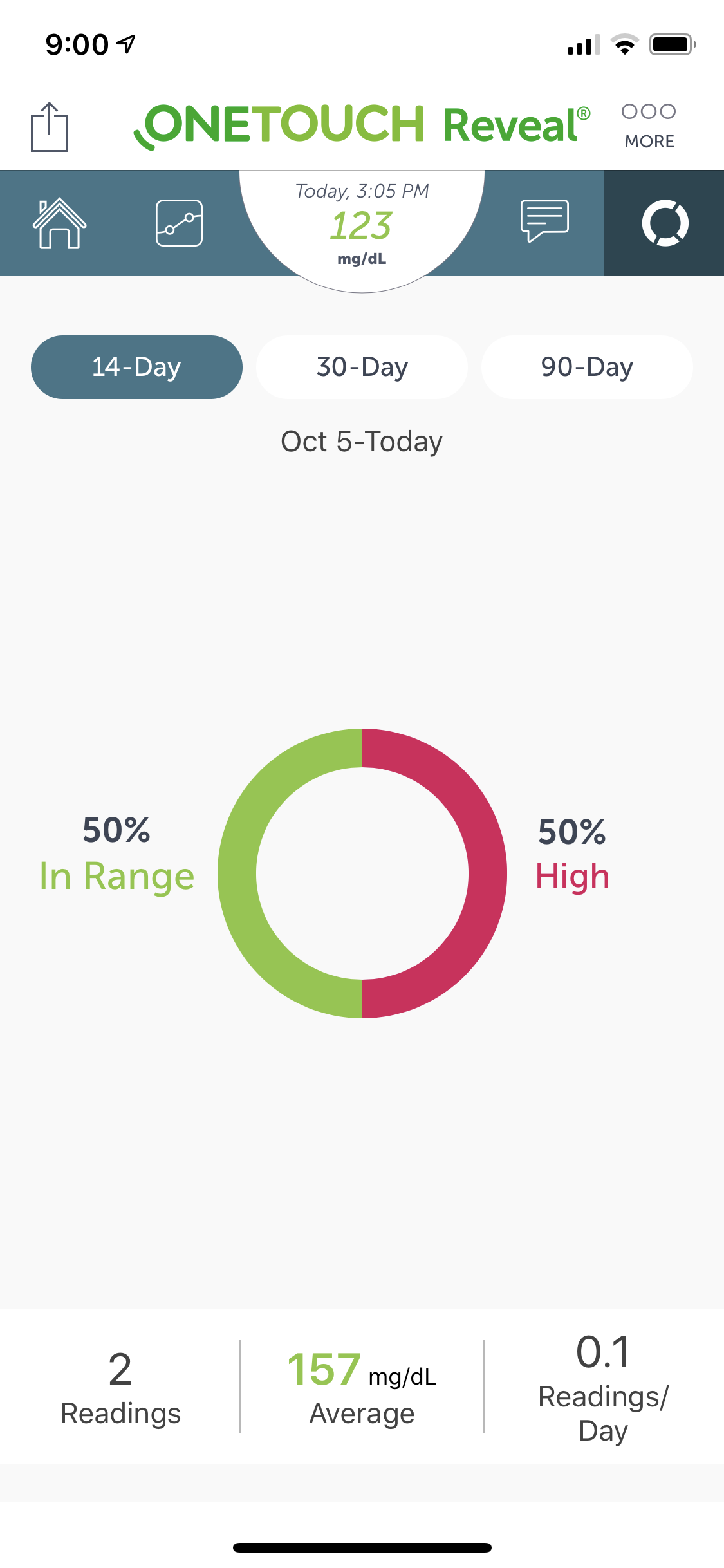
- Easily portable and highly customizable glucose monitoring for patients with diabetes. Real-time, dashboard informations provides a bolus calculator which provides precise insulin dosing.
- Because of the 510k submission to the FDA, the app, code and all services provided were scrutinized by the federal government to ensure safety and efficacy.
- The highly interactive and usable dashboard was designed to provide relevant and accurate real-time information. It was customizable and easily configurable.